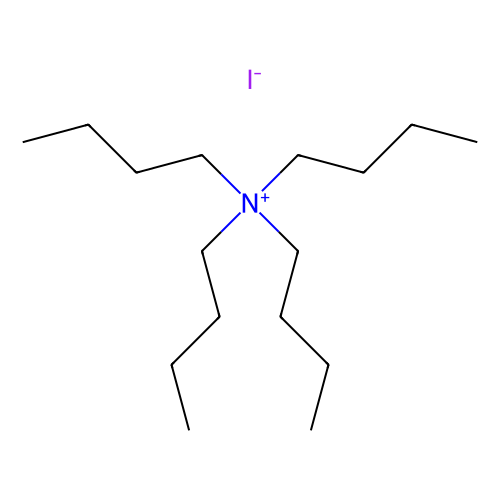
Tetrabutylammonium iodide (TBAI) has been used as a catalyst in the following reactions: · Synthesis of O-benzyl-N-Boc-L-tyrosine benzyl ester from N-Boc-L-tyrosine. · Conversion of 8-fluoro-1-aminonaphthalene into 1-(8-fluoro-naphthalen-1-yl)piperazine hydrochloride. · Synthesis of 1-(2,4-dichlorophenyl)-5-(4-(4-iodobut-1-ynyl)phenyl)-4-methyl-N-(piperidin-1-yl)-1H-pyrazole-3-carboxamide from 4-(4-(1-(2,4-dichlorophenyl)-4-methyl-3-(piperidin-1-ylcarbamoyl)-1H-pyrazol-5-yl)phenyl)but-3-yn-1-yl methanesulfonate. Other reactions where TBAI can be used as a catalyst: · TBAI-tert-butyl hydroperoxide system can catalyze the conversion of α-methyl styrene derivatives into allylic sulfones by reacting with sulfonylhydrazides under metal-free conditions. · Palladium(0)-catalyzed cross-coupling between benzylic zinc bromides and aryl or alkenyl triflates. · Three-component coupling of amines, carbon dioxide, and halides to form carbamates in the presence of cesium carbonate.
Specifications and Purity: ≥99%
MDL Number: MFCD00011636
Molecular Formula: C16H36NI
Molecular Weight: 369.37
EC Number: 206-220-5
PubChem CID: 67553
Isomeric SMILES: CCCC[N+](CCCC)(CCCC)CCCC.[I-]
Beilstein Registry Number: 3916152
Related Documents: https://aladdin-for-icloud-store.oss-cn-hangzhou.aliyuncs.com/aladdinsci/pdp/sds/1/T103714-SCI_8eefd4800eaef9b95cc29c82ebdda88b.pdf
- UPC:
- 12131507
- Condition:
- New
- HazmatClass:
- No
- WeightUOM:
- LB
- MPN:
- T103714-25g
- CAS:
- 311-28-4
- Product Size:
- 25g
- Hazard Statement Codes:
- H335:H319:H315:H302
- Precautionary Statement Codes:
- P305+P351+P338:P261

 akash.verma@cenmed.com
akash.verma@cenmed.com (732) 447-1115
(732) 447-1115