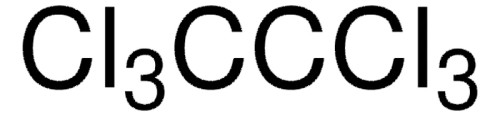General description
Effect of hydrogen peroxide concentrations on the degradation of hexachloroethane in the absence and the presence of tetrachloroethene by Fenton′s reagent has been investigated.
Application
Hexachloroethane was used as catalyst in preparation of chlorosilanes from hydrosilanes. It was used to investigate the transformation of hexachloroethane in natural waters. It was used as reagent during polycondensation reaction of p-aminobenzoic acid.
Molecular Weight: 236.74. Empirical Formula: C2Cl6. linearFormula: Cl3CCCl3. vapor density: 8.16 (vs air). Quality Level: 200. vapor pressure: 0.4 mmHg ( 20 °C). Assay: 99%. form: solid. mp: 183-185 . °C (dec.) (lit.). solubility: alcohol: soluble, benzene: soluble, chloroform: soluble, diethyl ether: soluble, oil: soluble, water: insoluble. density: 2.091 . g/mL . at 25 . °C (lit.). SMILES string: ClC(Cl)(Cl)C(Cl)(Cl)Cl. InChI: 1S/C2Cl6/c3-1(4,5)2(6,7)8. InChI key: VHHHONWQHHHLTI-UHFFFAOYSA-N. Pictograms: GHS07,GHS08,GHS09. Signal Word: Warning. Hazard Statements: H319 - H351 - H410. Precautionary Statements: P202 - P264 - P273 - P280 - P305 + P351 + P338 - P308 + P313. Hazard Classifications: Aquatic Acute 1 - Aquatic Chronic 1 - Carc. 2 - Eye Irrit. 2. Storage Class Code: 11 - Combustible Solids. WGK: WGK 3. Flash Point(F): Not applicable. Flash Point(C): Not applicable. Personal Protective Equipment: Eyeshields, Gloves, type P3 (EN 143) respirator cartridges.- UPC:
- 12170000
- Condition:
- New
- HazmatClass:
- No
- MPN:
- 185442-5G
- CAS:
- 67-72-1

 akash.verma@cenmed.com
akash.verma@cenmed.com (732) 447-1115
(732) 447-1115