Analysis Note
Assay (acidimetric): ≥ 99.7 %
Color: ≤ 10 Hazen
Titratable base: ≤ 0.0004 meq/g
Acetic anhydride: ≤ 100 ppm
Chloride (Cl): ≤ 1 ppm
Heavy metals (as Pb): ≤ 0.5 ppm
Sulfate (SO₄): ≤ 1 ppm
Fe (Iron): ≤ 0.2 ppm
Substances reducing potassium dichromate: passes test
Substances reducing potassium permanganate: passes test
Evaporation residue: ≤ 10 ppm
Dilution test: passes test
Legal Information
EMPARTA is a registered trademark of Merck KGaA, Darmstadt, Germany
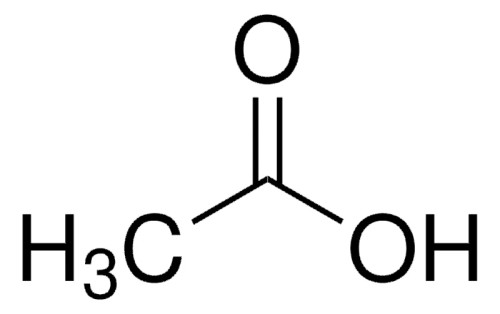
Molecular Weight: 60.05. Empirical Formula: C2H4O2. linearFormula: CH3CO2H. grade: ACS reagent. Quality Level: 200. vapor density: 2.07 (vs air). vapor pressure: 15.4 hPa ( 20 °C). product line: EMPARTA®. . Assay: ≥. 99.7% (acidimetric). form: liquid. autoignition temp.: 485 . °C, 800 . °F. potency: 3310 . mg/kg LD50, oral (Rat). expl. lim.: 16 . %, 92 . °F, 4 . %, 59 . °F. impurities: ≤. 0.0004 . meq/g Titratable base, ≤. 100 . ppm Acetic anhydride. evapn. residue: ≤. 10 . ppm. color: APHA: ≤. 10. refractive index: n- UPC:
- 41105317
- Condition:
- New
- HazmatClass:
- Yes
- MPN:
- 1018302500
- CAS:
- 64-19-7

 akash.verma@cenmed.com
akash.verma@cenmed.com (732) 447-1115
(732) 447-1115